The short answer: when doctors and public health officials report bird flu mortality rates, they are almost always talking about deaths among confirmed, lab-tested cases. That group skews heavily toward the sickest people, which is exactly why the numbers look so alarming. For H5N1, the most-watched strain, the cumulative case fatality ratio sits around 60% based on WHO-tracked laboratory-confirmed cases going back to 2003. That figure does not mean six out of ten people who catch H5N1 die. It means that among people sick enough to get tested and confirmed, roughly six in ten have died. That distinction matters enormously, and the rest of this article unpacks why the rate is still genuinely high even after accounting for that bias, and what you can actually do about it.
Why Is Bird Flu Mortality Rate So High? Key Reasons

What 'bird flu mortality rate' actually means
The term you will see in news reports is case fatality ratio, or CFR. It is calculated by dividing confirmed deaths by laboratory-confirmed cases. WHO tracks this for every major bird flu subtype, publishing cumulative tables that update as cases are reported. The CFR is not the same as infection fatality rate, which would count everyone infected including people with mild or no symptoms who were never tested. Because bird flu surveillance mostly catches severely ill people, the CFR overstates risk for a random person exposed to infected poultry, but it does accurately describe how deadly the virus is for the people who get sick enough to come to medical attention.
CFR numbers also shift during an outbreak. WHO noted this clearly with H7N9: the CFR among confirmed cases was reported at around 22% as of January 31, 2014, early in that outbreak's course. As more cases were identified and outcomes tracked, later analyses put the overall H7N9 case fatality ratio closer to 37%. That movement happened not because the virus changed but because the denominator (total confirmed cases) and the time needed for deaths to be recorded kept adjusting. So when you read a mortality figure for bird flu, always ask: from which point in the outbreak, and among which population?
Why the virus itself drives such high fatality

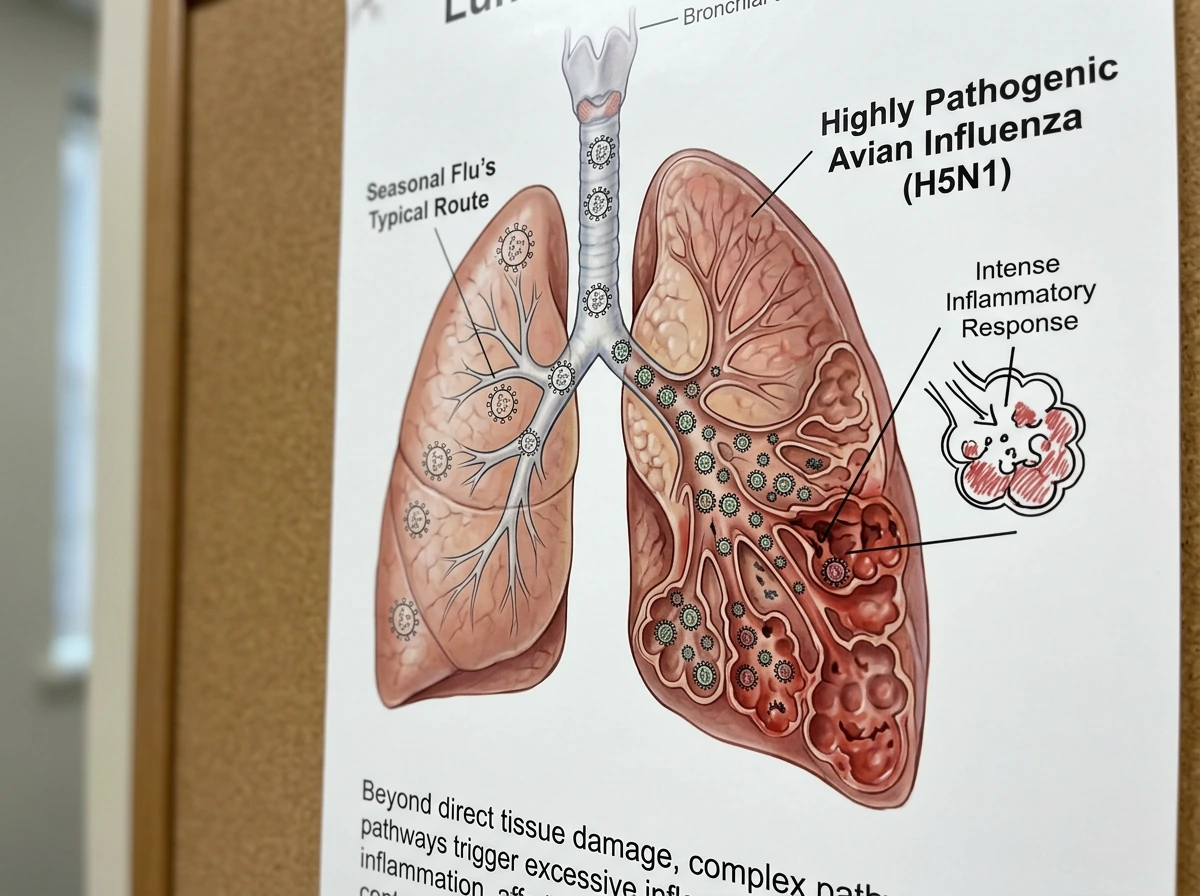
Highly pathogenic avian influenza viruses, particularly H5N1 and H5N6, behave differently in the human body than seasonal flu does. Seasonal flu mainly infects the upper respiratory tract, which is uncomfortable but manageable for most people. HPAI viruses bind more efficiently to receptors deeper in the lungs, which means they cause severe viral pneumonia more often and more quickly. That pneumonia can progress to acute respiratory distress syndrome (ARDS), a state where the lungs fill with fluid and oxygen exchange collapses. Most hospitalized human H7N9 patients, for example, developed viral pneumonia, and a significant portion progressed to ARDS. This is not a mild complication. ARDS typically requires mechanical ventilation and intensive care.
Beyond the mechanical damage to the lungs, highly pathogenic strains trigger an exaggerated immune response. The body essentially overreacts, releasing inflammatory signals that damage healthy tissue alongside infected cells. This cytokine storm effect has been documented in H5N1 cases and contributes to multi-organ failure in severe cases. The combination of deep lung infection and immune dysregulation is what makes these strains genuinely lethal, not just in frail patients but sometimes in otherwise healthy adults.
How transmission dynamics and detection timing inflate the numbers

Bird flu does not spread easily between people right now. Nearly all human cases involve direct or close unprotected contact with infected birds or contaminated environments. That means the people who get infected are often farmworkers, live-poultry market workers, or people in rural areas where access to rapid medical care can be limited. By the time someone with H5N1 gets to a hospital, gets tested, and gets confirmed, the disease may already be advanced. Delayed arrival to care is one of the strongest contributors to the high observed mortality rate.
Surveillance design also plays a role. WHO explicitly notes that most human cases are detected through severe-illness surveillance, meaning the system is designed to catch the worst cases. People with genuinely mild bird flu infections, if they exist in meaningful numbers, are largely invisible to these tracking systems. That surveillance bias pulls the reported CFR upward. It does not mean the virus is safe, but it does mean the true population-level risk is not as precisely described by the CFR as it might appear.
Strain-by-strain breakdown: not all bird flu is equal
One of the most practically useful things to understand is that 'bird flu' covers several distinct subtypes with very different fatality profiles. Here is how the major strains compare based on WHO and peer-reviewed data.
| Strain | Pathogenicity | Approximate CFR (confirmed cases) | Primary clinical pattern |
|---|---|---|---|
| H5N1 | Highly pathogenic (HPAI) | ~60% (WHO cumulative, 2003–2024) | Severe viral pneumonia, ARDS, multi-organ failure |
| H5N6 | Highly pathogenic (HPAI) | ~66% (per comparative epidemiology studies) | Similar to H5N1; severe lower respiratory disease |
| H7N9 | High pathogenicity variant exists; initial strain was LPAI | ~37% (later outbreak estimates); ~22% at early reporting | Viral pneumonia, ARDS in severe cases; wider spectrum of severity |
| H9N2 | Low pathogenicity (LPAI) | Very low; most cases mild | Upper respiratory illness, conjunctivitis; rarely severe |
The contrast between HPAI and LPAI strains is striking. A comparative epidemiology study found that highly pathogenic strains like H5N1 and H5N6 had a combined CFR of 66%, while low pathogenicity strains like H7N9 (in its earlier, lower pathogenicity form) and H9N2 had substantially lower fatality. The biology explains this: HPAI strains replicate more aggressively and spread more readily to tissues beyond the respiratory tract. LPAI strains, by contrast, tend to stay in the upper airways and cause milder illness, similar to a bad cold or mild flu.
H7N9 sits in an interesting middle ground. It started as a low pathogenicity strain in poultry but caused severe human disease, and a highly pathogenic variant emerged during later outbreak waves. Its CFR, while lower than H5N1, is still far above seasonal flu, which kills roughly 0.1% of confirmed cases in a typical year. The practical takeaway: when news reports mention bird flu, knowing which strain is circulating tells you a lot about how seriously to treat the risk.
How diagnosis, treatment timing, and access to care change outcomes
This is the part of the mortality picture that is most actionable. The antiviral drug oseltamivir (Tamiflu) is the first-line treatment for bird flu, and it works, but timing is everything. CDC is explicit that earlier oseltamivir treatment is associated with better survival than later or no treatment. Research on hospitalized influenza pneumonia patients shows that starting oseltamivir within 48 hours to 5 days of illness onset improves survival, and treatment initiated within 24 hours can make a significant difference in mortality outcomes. Wait too long and the virus has already caused enough damage to the lungs that an antiviral has little to work with.
The challenge in real-world outbreaks is that bird flu is rare enough that clinicians may not immediately recognize it. Someone comes in with fever, cough, and shortness of breath, and the first assumption is seasonal flu or pneumonia. If the patient does not volunteer information about recent poultry exposure, testing for novel influenza A may be delayed. By the time the lab confirms H5N1, critical treatment hours have passed. This diagnostic lag is a structural contributor to high mortality in detected cases.
CDC addresses this directly. Current guidance instructs clinicians not to wait for laboratory confirmation before starting oseltamivir in a patient who meets exposure criteria (contact with infected or potentially infected poultry) and has compatible respiratory symptoms. The recommendation is to initiate empiric antiviral treatment as soon as possible, even before test results are back. For hospitalized patients with confirmed, probable, or suspected HPAI A(H5N1), antivirals should be started regardless of how long ago symptoms began. That is how seriously timing is taken.
Beyond antivirals, survival in severe cases depends on the quality of intensive care available. ARDS requires ventilator support, prone positioning, and careful fluid management. Those resources are readily available in well-equipped hospitals but may be limited in rural settings or lower-income countries where many bird flu exposures occur. This access gap helps explain why mortality differs between outbreaks in different countries, even for the same strain.
Your actual risk right now, and what to do about it
For most people reading this in March 2026, the personal risk of contracting bird flu is low. The virus does not spread easily from person to person, and the overwhelming majority of cases involve people who had direct, unprotected contact with infected birds or their environments. That said, 'low risk' is not 'no risk,' and the steps that reduce risk are simple.
Check your exposure level first
CDC defines exposure categories to help assess risk. High-risk exposures include unprotected direct contact with sick or dead birds known or suspected to have bird flu, handling infected animals without appropriate PPE, or working in a facility with a confirmed outbreak. Lower-risk exposures involve being near (but not in direct contact with) birds in an affected area while using appropriate precautions. Knowing which category applies to you determines what monitoring and medical steps are warranted.
Symptoms that mean go to the doctor today
If you have had a relevant exposure (poultry contact, visit to a live bird market, work in an affected agricultural setting) and develop any of the following within 10 days, seek medical care immediately and tell the clinician about your exposure before they start testing:
- Fever (or feeling feverish) combined with cough, sore throat, or runny nose
- Shortness of breath or difficulty breathing
- Chest pain or tightness
- Altered mental status or confusion
- Conjunctivitis (eye redness and discharge) alongside respiratory symptoms
Do not downplay the exposure history when you talk to your doctor. That information is what triggers the right testing protocol and, critically, empiric antiviral treatment before confirmation. Every hour matters once you are symptomatic and have had a real exposure.
Post-exposure steps if you think you were exposed
CDC recommends post-exposure prophylaxis with oseltamivir for close contacts of a confirmed or probable HPAI A(H5N1) patient, with a standard course of twice-daily dosing for five days. If you have had a close, unprotected exposure to a confirmed human case (not just birds), contact your local or state health department right away. They will assess whether prophylaxis is appropriate and coordinate with you on monitoring.
Practical prevention for high-risk occupations
- Wear appropriate PPE (gloves, eye protection, N95 respirator) when handling birds or working in environments with potential bird flu exposure
- Avoid touching your face, eyes, or mouth with ungloved hands near birds or their waste
- Stay current on seasonal flu vaccination: it does not protect against bird flu, but it reduces confusion with seasonal illness and may lower the risk of co-infection
- Know the reporting chain: if you work in agriculture or veterinary medicine, know who to call if you observe unusual bird deaths or illness in your flock
- Monitor for symptoms for 10 days after any significant poultry exposure, even if you felt fine during the exposure
Where to get current information

The situation with bird flu strains and active outbreaks changes. CDC and WHO both maintain current guidance pages for HPAI A(H5N1) and other subtypes that are updated as new data comes in. Before traveling to regions with active poultry outbreaks or if you work in an agricultural setting, check the CDC Health Alert Network (HAN) notices and WHO's influenza at the human-animal interface summaries. These are the same sources clinicians use and are written clearly enough for non-specialists to follow.
The bottom line on why bird flu mortality rates are so high comes down to three overlapping factors: a virus that attacks deep lung tissue and causes the immune system to overreact, a detection system that mostly finds the sickest patients, and delays in diagnosis and treatment that give the virus time to do serious damage before antivirals are started. All three of those factors are real, but the third one is the most actionable. This is why is bird flu so deadly, and it also connects directly to how CFR can look high in the confirmed cases that get detected. If you have had an exposure and get sick, say so immediately, get tested fast, and push for empiric antiviral treatment while results are pending. So, is the bird flu deadly? Yes, that is why the question <a data-article-id="F18F790B-A994-48E4-A8C8-53492418A46B"><a data-article-id="F18F790B-A994-48E4-A8C8-53492418A46B"><a data-article-id="F18F790B-A994-48E4-A8C8-53492418A46B"><a data-article-id="F18F790B-A994-48E4-A8C8-53492418A46B">why is bird flu so deadly</a></a></a> matters, because the same three factors explain both the CFR and what you can do once you might be exposed. Dogs can also get infected with bird flu viruses, but serious illness or death in dogs is uncommon and depends on the specific virus and exposure So, is the bird flu deadly?. </a> That is why the case fatality ratio can look very high in confirmed cases, even though most people have a low chance of getting infected how deadly is bird flu. In the specific groups that get confirmed and treated late, reported fatality can look very high, but your personal risk depends on exposure and how quickly you get care get tested fast. That window of early treatment is where lives are saved.
FAQ
If a bird flu CFR is 60%, does that mean 60% of people who get exposed will die?
Not automatically. Published figures are usually case fatality ratios for lab-confirmed, symptomatic patients, not “everyone exposed” risk. If the report does not clearly state the subtype, the outbreak time window, and whether it is based on confirmed cases only or also probable cases, the comparison to your personal risk can be misleading.
Why can the mortality rate look higher when bird flu is rare?
A key reason the CFR can look worse is missing mild or asymptomatic infections. If surveillance primarily recruits people with severe symptoms, the denominator shrinks while the numerator stays visible, which inflates the CFR even if the virus produces a broader range of illness in the general exposed population.
Can the mortality number change after an outbreak is “over”?
Yes, timing can matter for the same patient group. Some CFR calculations are “cumulative” but still incomplete while patients are still being followed, or they can update as late deaths are recorded. That is why later analyses can report a different CFR than early outbreak reports.
Why do bird flu mortality numbers differ so much between news stories?
The most relevant marker is not the overall label “bird flu,” but the subtype and whether it is described as highly pathogenic or low pathogenic. Different strains have different lung involvement and typical severity, so mixing subtypes under one headline can hide that some are lower risk than others.
What does it mean clinically to start treatment before lab results are back?
Empiric treatment is about “meeting exposure and symptom criteria,” not about waiting for lab confirmation. If a clinician sees compatible respiratory symptoms plus a plausible exposure history, guidance generally supports starting oseltamivir promptly because outcomes worsen when antivirals are delayed.
If symptoms started days ago, is oseltamivir still useful?
Earlier treatment can improve survival, but clinicians also consider severity and how long symptoms have been present. In real-world settings, some patients may present late, and while antivirals may still be given, the benefit is smaller once severe lung injury has progressed. That is why the recommended framing focuses on starting as soon as possible within the effective window.
How does exposure history affect diagnosis and survival?
Testing delay is only one piece. Clinicians also need to think about differential diagnosis, such as seasonal influenza, bacterial pneumonia, or other respiratory viruses. If the exposure history is not volunteered, the initial probability estimate can stay low, which can delay both the correct antiviral plan and the right level of respiratory monitoring.
Does ICU or hospital resources change the mortality rate even for the same strain?
Mortality is not just the virus, it is also the care environment. Severe cases often require ventilatory support, ARDS management, and close monitoring for multi-organ failure. Hospitals with limited ICU capacity, delayed access to oxygen or ventilation, or fewer supportive-care resources can see higher observed deaths even when the strain is similar.
What should I do if I had an exposure but it was not “direct contact”?
Lower-risk exposure does not always mean you should ignore symptoms. If you had an exposure that meets recognized categories (for example, unprotected direct contact with sick or dead birds), and you develop compatible symptoms within the specified post-exposure window, you should seek medical care urgently and mention the exposure immediately.
Who actually qualifies for post-exposure prophylaxis with oseltamivir?
Post-exposure prophylaxis is targeted to certain close contacts in specific scenarios, such as close, unprotected contact with a confirmed human H5N1 case or other guidance-based criteria. It is not generally intended for casual contact with birds or routine travel through an affected area.
How fast should I seek care after poultry or market exposure if I feel sick?
Practical triage should be driven by the combination of exposure timing and symptom onset. If you develop fever and respiratory symptoms within the relevant window after exposure, you should seek care right away and disclose the exposure before testing decisions are made, because it can change whether empiric antivirals are started.
Can I treat bird flu risk the same way as seasonal flu precautions?
Yes. “Bird flu” can be mistaken for seasonal flu risk. Seasonal influenza is far more transmissible person to person, while current bird flu events involve limited human-to-human spread. Using seasonal-flu assumptions can lead to underestimating the importance of exposure history.

